Uterine fibroid: what you need to know
- Frequency. Up to 70-80% of women develop a uterine fibroid during their lifetime, with peak incidence between ages 40 and 50.
- Symptoms that warrant consultation. Heavy periods, pelvic pain, urinary discomfort, infertility — an asymptomatic fibroid does not require treatment.
- 3 main strategies. Myomectomy (preserves uterus, first line if pregnancy planned), hysterectomy (definitive solution), embolisation (no surgery).
- Decisive criterion. Pregnancy plans, age, and fibroid mapping guide the choice — no universal treatment.
- Timeline. No absolute urgency — 1 to 3 months to organise surgery after a complete workup.
- Recurrence. 10-30% at 5 years after myomectomy; 0% after hysterectomy; 15-25% at 5 years after embolisation.
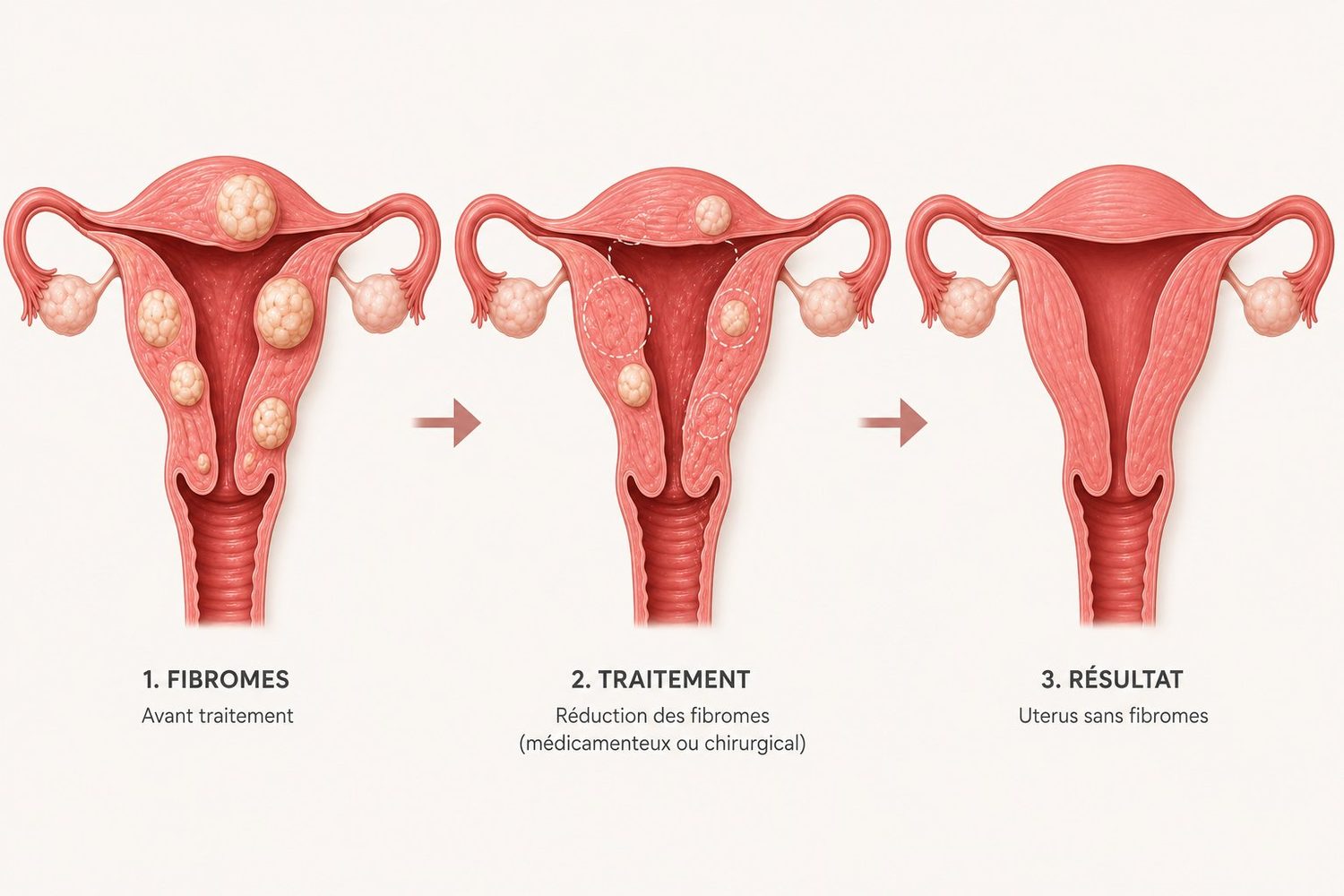
Uterine fibroids are among the most common gynecologic conditions: an estimated 70 to 80% of women will develop them at some point in their lives, with peak incidence between ages 40 and 50. These benign tumours of the uterine smooth muscle remain entirely silent in many women. But when they become symptomatic — heavy bleeding, pelvic pain, urinary or bowel discomfort, fertility issues — the question of treatment arises.
And with it, a central question: do you actually need surgery? And if so, which technique? Conservative myomectomy (removing only the fibroid), hysterectomy (removing the entire uterus), uterine artery embolization (cutting off the fibroid's blood supply)? Each option has its indications, advantages and limitations. This article reviews the three strategies to help you understand the parameters of the decision. For the broader surgical management of uterine conditions, also see our page on benign uterine surgery.
Understanding uterine fibroids
Uterine fibroids — also called leiomyomas or myomas — are benign tumours that grow from the muscle cells of the uterine wall (this muscular wall is called the myometrium). Their growth is sensitive to female hormones, particularly oestrogen and progesterone, which explains their typical course: they appear and grow during the reproductive years, then stabilise or shrink at menopause.
An anatomical classification that guides strategy
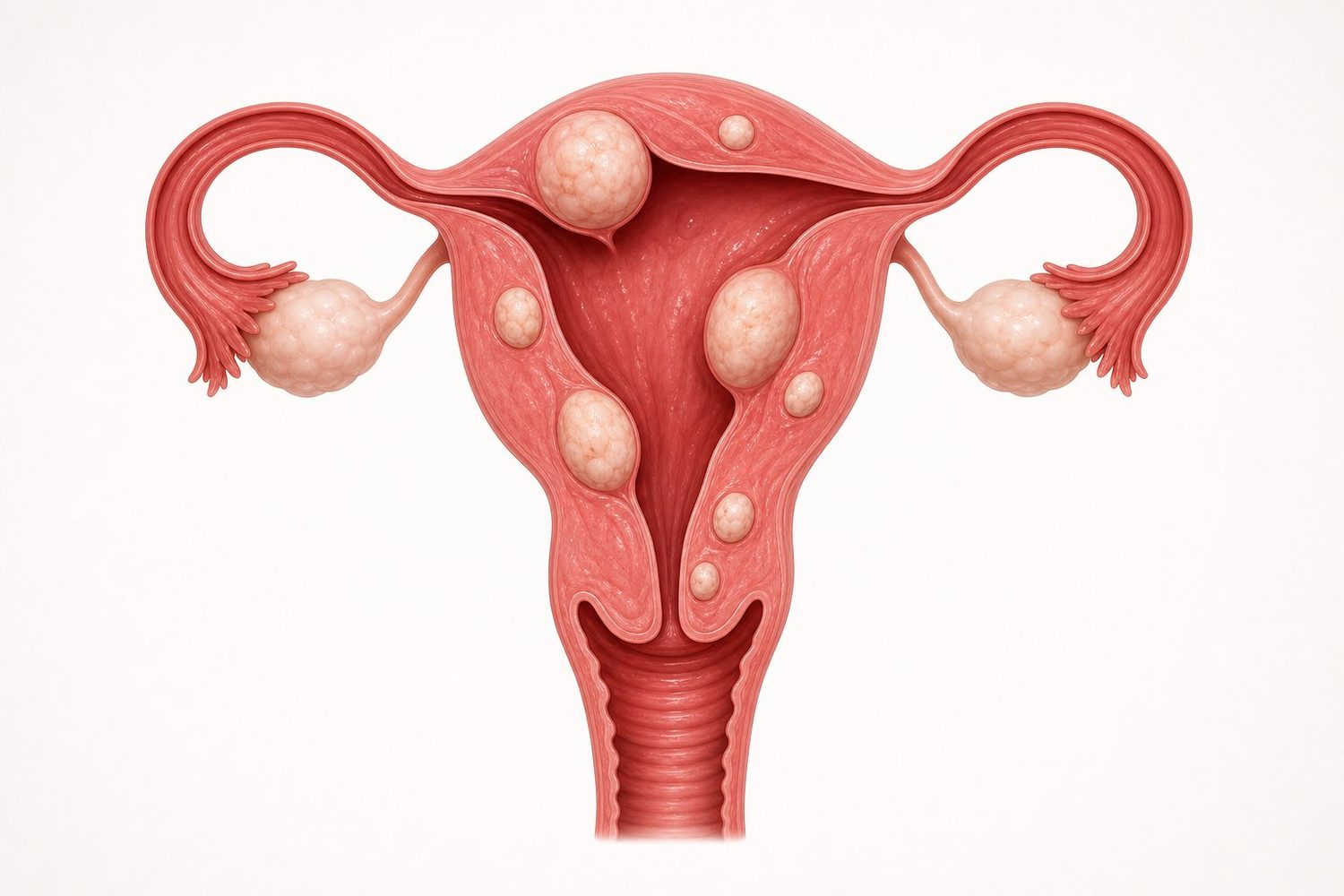
Where do fibroids grow inside the uterus?
Fibroids can develop at different locations within the muscular wall of the uterus. Location matters enormously: a small fibroid bulging into the uterine cavity may cause heavy bleeding, while a large one growing on the outer surface can stay completely silent.
This is why precise imaging is essential before any treatment decision: a fibroid is not managed the same way depending on its position.
The location of a fibroid in the uterine wall directly determines symptoms and treatment options. The international FIGO classification distinguishes 8 types, grouped into four broad families:
- Submucosal fibroids (FIGO 0, 1, 2): they bulge into the cavity of the uterus, where the embryo would implant. They are the main cause of heavy bleeding and conception difficulties. They can be removed via the natural route, through the cervix (hysteroscopy).
- Intramural fibroids (FIGO 3, 4): located within the thickness of the uterine muscle itself, without reaching the inner cavity or the outer envelope. Symptoms vary.
- Subserosal fibroids (FIGO 5, 6): they grow outward from the uterus, bulging beneath the outer envelope. They mostly cause pressure symptoms (bladder, rectum) and a feeling of pelvic heaviness.
- Pedunculated fibroids (FIGO 7): connected to the uterus by a thin "stalk", like a fruit hanging from its branch. There is a rare but real risk of sudden torsion, which can trigger acute pain.
This map is essential: a 3 cm submucosal fibroid can be far more symptomatic than an 8 cm subserosal one. It is the position, as much as the size, that drives strategy.
Worth remembering. Having a uterine fibroid is not a disease in itself. Most fibroids will never cause symptoms and never need treatment. It is the appearance of symptoms — and their impact on your daily life — that shifts things into a treatment logic. To explore all benign uterine conditions managed at the practice, see the dedicated page.
Pelvic ultrasound, the starting point of the work-up
Ultrasound is the cornerstone of diagnosis. Performed both through the abdomen and with an endovaginal probe, it is entirely painless and allows the uterus to be visualised in just a few minutes — counting fibroids, measuring their size, defining their position.
This first test often shapes the whole strategy: simple monitoring, medical treatment or surgery.
The work-up: ultrasound, MRI, hysteroscopy
Reading the images, a step of its own
The scans are reviewed and interpreted by a specialist radiologist, who measures each fibroid, defines its position using the FIGO classification, and characterises its appearance. This detailed report is then discussed in consultation to design the right treatment strategy.
When doubt persists — rapid growth, atypical MRI features — a second radiology opinion can be requested to confirm the analysis.
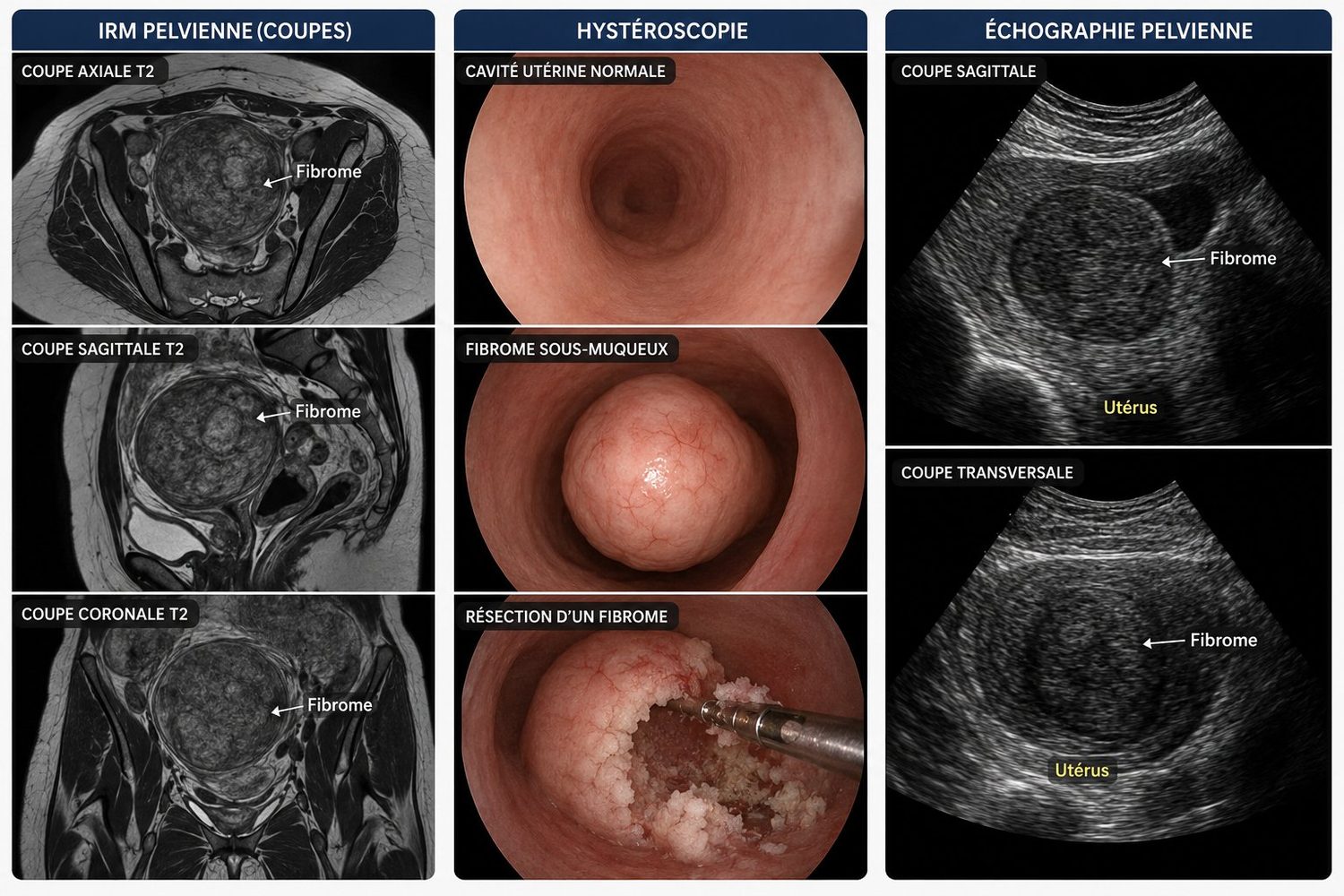
Before any treatment decision, precise imaging is essential. It rests on three pillars:
- Pelvic ultrasound via abdominal and endovaginal routes (a probe inserted into the vagina): the first-line examination, painless and inexpensive. It allows fibroids to be counted, measured and located.
- Pelvic MRI: indicated when the uterus contains many fibroids or very large fibroids, or before complex treatment. It provides a complete map and accurately characterises each fibroid.
- Diagnostic hysteroscopy: a thin optical tube inserted via the natural route allows direct visualisation inside the uterine cavity. Particularly useful when a submucosal fibroid is suspected.
Blood tests complete the assessment: a full blood count to measure any anaemia caused by bleeding, iron studies (ferritin) and, depending on context, hormonal tests and full preoperative work-up.
When to treat a uterine fibroid?

Symptoms that impact daily life
Many women live with a uterine fibroid without even knowing it. But when periods become too heavy, pain sets in or pressure builds on the bladder or bowel, quality of life can be significantly affected — and that impact is precisely what justifies a surgical opinion.
The treatment decision is not based on the existence of the fibroid itself, but on its clinical consequences. The main situations that justify treatment are:
- Heavy and prolonged periods, or bleeding between periods, which eventually cause exhaustion (anaemia with low iron levels).
- Chronic pelvic pain or very painful periods that disrupt daily life.
- Pressure on neighbouring organs: frequent need to urinate, sense of incomplete bladder emptying, constipation or pelvic heaviness.
- Infertility attributed to the fibroid after a full work-up of the couple.
- Rapid growth of the fibroid, particularly after menopause, prompting reconsideration.
- Significant uterine volume with visible abdominal protrusion.
Conversely, an asymptomatic fibroid — even a large one — can simply be monitored by yearly ultrasound. Many post-menopausal patients see their fibroids shrink spontaneously due to the hormonal drop.
Medical treatment: sometimes enough on its own
Before considering an invasive procedure, several medical options can be offered: progestogens, levonorgestrel intrauterine device, GnRH agonists, ulipristal acetate (under strict prescribing restrictions). These are mainly useful to reduce bleeding and fibroid volume before surgery, or as an alternative for patients close to menopause.
Caught between medical treatment and surgery?
Dr Zeitoun designs a tailored strategy with you, based on your age, symptoms, pregnancy plans and the map of your fibroids.
Myomectomy: preserving the uterus
Myomectomy, removing the fibroid without touching the uterus
The aim: remove the fibroid (or fibroids) and rebuild the uterine wall so it regains its strength and shape. This is the reference option for younger women or those with pregnancy plans.
Depending on the position and size of the fibroids, surgery is performed via the natural route, through small abdominal incisions, or — less commonly — through a wider incision.
Myomectomy involves surgical removal of the fibroid(s) while preserving the uterus. It is the reference treatment for women wishing to preserve fertility or to keep their uterus for personal reasons. Several surgical approaches are available, chosen based on the fibroid map.
Available techniques
- Hysteroscopic myomectomy: indicated for submucosal fibroids (FIGO 0, 1, 2). A minimally invasive technique through the natural route, scarless, usually as an outpatient. It effectively treats heavy bleeding caused by a fibroid bulging into the cavity.
- Laparoscopic myomectomy: minimally invasive technique through small abdominal incisions. Preferred for moderate-sized intramural or subserosal fibroids in limited numbers. Quick recovery, short hospital stay (24 to 48 hours), minimal scarring.
- Open myomectomy (laparotomy): open route via an abdominal incision, reserved for complex cases — multiple fibroids, very large fibroids, significant prior surgical history. Allows better intraoperative bleeding control and a sturdy suture of the uterine wall — essential if pregnancy is planned afterwards.
- Robot-assisted myomectomy: a laparoscopic variant, useful for complex sutures, but with no proven long-term benefit over standard laparoscopy.
Outcomes and limitations
Myomectomy provides excellent symptom control: improvement of heavy periods in 80 to 90% of cases, reduction of pelvic pain in most patients, and preserved fertility with pregnancy rates comparable to women without fibroids in published data. The main drawback is the risk of recurrence, estimated at 10 to 30% at 5 years. Two reasons explain this: fibroids are hormone-sensitive and can re-emerge until menopause, and very small fibroids may go undetected at the first surgery.
And what about vaginal delivery afterwards? The answer depends on how extensive the surgery was on the uterine muscle. When myomectomy involved a significant opening of the uterine wall to remove a large fibroid, the surgeon may recommend a scheduled caesarean section for the next pregnancy. The reasoning is straightforward: during labour contractions, the uterus is heavily strained, and the muscle scar could theoretically give way — this is called uterine rupture. The risk is rare but justifies caution. By contrast, after a simple resection via the natural route (hysteroscopy) or removal of a small surface fibroid, vaginal delivery remains entirely possible. The decision is always made case by case, in consultation between the surgical and obstetric teams.
The recommended interval between myomectomy and a new conception is usually 6 months — the time needed for the uterine muscle to heal solidly.
Hysterectomy: a definitive solution
Hysterectomy, turning the page for good
When fibroids are too numerous or too large, or when bleeding doesn't yield to any treatment, removing the uterus altogether ends symptoms — and recurrence — definitively.
Surgery is most often performed laparoscopically, sometimes via the natural route. The ovaries are kept whenever possible.
Hysterectomy involves removing the uterus. It is the definitive treatment for uterine fibroids, with no risk of recurrence. It is mainly offered to women no longer wishing to become pregnant, in situations where conservative treatments have failed, or for multiple fibroids that make myomectomy unrealistic. For the technical details of the surgery itself, see the dedicated page on hysterectomy.
Surgical approaches
| Type | Approach | Main indications | Advantages |
|---|---|---|---|
| Total laparoscopic hysterectomy | Minimally invasive (4 ports) | Multiple fibroids, uterus < 16 weeks size | Quick recovery, minimal scars |
| Vaginal hysterectomy | Natural route | Mobile uterus, associated prolapse | No abdominal scar |
| Open hysterectomy (laparotomy) | Open route | Very large uterus, significant adhesions | Better control in complex cases |
| Subtotal hysterectomy | Mostly laparoscopic | When cervix preservation is desired | May preserve vaginal anatomy |
The choice of approach depends on uterine volume, prior surgical history, uterine mobility and any associated condition. Laparoscopy is now favoured whenever technically possible.
The question of the ovaries
Hysterectomy for fibroids does not necessarily involve removal of the ovaries. In a non-menopausal woman without a specific risk factor (notably no BRCA mutation), preserving the ovaries is the rule — to maintain hormonal function and avoid early surgical menopause. The decision to remove or preserve the ovaries is made case by case, based on age, family history and overall context. To understand ovarian and tubal surgery as a whole, see the page on tubes and ovaries.
Benefits and drawbacks
The benefits are clear-cut: complete and definitive symptom resolution, no risk of recurrence, end of periods, lasting improvement in quality of life, and even reduced endometrial cancer risk. Patient satisfaction after hysterectomy for symptomatic fibroids is high in the literature.
The drawbacks need to be addressed openly: definitive end of fertility, sometimes underestimated psychological impact, surgical risks inherent to any abdominal procedure (vascular, ureteral or bowel injury, infection, thrombosis), and more rarely, changes in sexual or pelvic function afterwards. Honest, complete information at the preoperative visit is essential.
Uterine artery embolization: a minimally invasive alternative
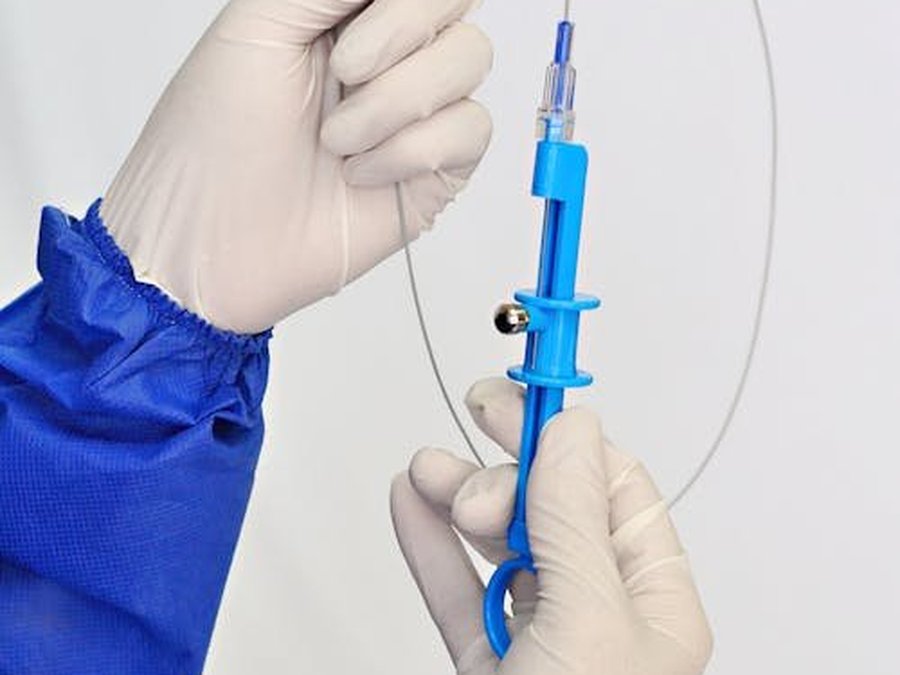
Embolization, cutting off the fibroid's blood supply
No scalpel, no opening, no general anaesthesia. A specialised radiologist threads a fine catheter through the groin and blocks the vessels that feed the fibroid — which then shrinks gradually over several months.
A valuable alternative for women who refuse surgery or cannot have it.
Uterine artery embolization (UAE, also called UFE in the US) is an interventional radiology technique performed by a specialised radiologist. It is not surgery in the classic sense: it is a percutaneous procedure done through a small puncture in the groin, under local anaesthesia or light sedation (sometimes paired with mild analgesia for comfort).
How it works
Under radiological guidance, the radiologist threads a fine catheter (a soft tube) into the arteries that bring blood to the uterus, and injects tiny biocompatible microspheres. These microspheres block blood flow to the fibroids, which — deprived of their blood supply — shrink gradually over several months. The healthy part of the uterine wall, better supplied by other small vessels, is largely spared.
Advantages and limitations
| Criterion | Advantages | Limitations |
|---|---|---|
| Invasiveness | Percutaneous procedure, no surgery | Ionising radiation exposure |
| Hospital stay | Day case or 24 to 48 hours | Post-embolization syndrome (pain, fever) |
| Recovery | Return to activity in 1 to 2 weeks | Sometimes marked pelvic pain |
| Uterine preservation | Uterine anatomy preserved | Possible impact on ovarian reserve |
| Recurrence | — | Re-intervention in 15 to 25% at 5 years |
Who is it for?
Embolization is an excellent option for patients who refuse surgery, have anaesthetic contraindications, or wish to preserve their uterus without immediate pregnancy plans. It is more controversial in women planning a pregnancy: most international societies recommend myomectomy as first-line in this case, due to uncertainties about subsequent fertility and obstetric outcomes.
Pure submucosal fibroids or pedunculated fibroids are situations where embolization is less suitable. By contrast, uteruses with many fibroids in women close to menopause are an excellent indication.
Important. Embolization is a collaborative decision between the patient, the gynecologist and the interventional radiologist. A pre-procedure planning meeting with pelvic MRI is systematic to confirm the indication and anticipate the expected outcome.
How to choose between the three options?

None of the three techniques is universally superior: the right treatment is the one that fits your personal situation. Several parameters drive the decision:
- Age and proximity to menopause: the closer to menopause, the more relevant a conservative or minimally invasive approach becomes, since fibroids shrink spontaneously after periods stop.
- Pregnancy plans: decisive. Pregnancy desired → myomectomy first-line. No pregnancy plans → all options remain open.
- Fibroid map: number, size, position. A single fibroid bulging into the uterine cavity is treated via the natural route (hysteroscopy), whereas a uterus with many fibroids may better suit hysterectomy or embolization.
- Severity of symptoms: severe anaemia, badly impaired quality of life, failed medical treatment all point towards a radical or definitive strategy.
- Comorbidities and anaesthetic risk: may tip the balance towards a less invasive approach.
- Personal preferences: how you feel about your uterus, willingness to accept the risk of recurrence, attitude towards the surgical act itself.
Comparative summary
| Myomectomy | Hysterectomy | Embolization | |
|---|---|---|---|
| Uterine preservation | Yes | No | Yes |
| Pregnancy compatible | Yes | No | Debated |
| Recurrence possible | 10 to 30% at 5 years | None | 15 to 25% at 5 years |
| Anaesthesia | General | General | Local or light sedation |
| Hospital stay | 1 to 5 days | 1 to 5 days | Day case or 24 hr |
| Return to activity | 2 to 4 weeks | 3 to 6 weeks | 1 to 2 weeks |
| Induced menopause | No | No if ovaries preserved | Possible (rare) |
The aim of the surgical consultation is precisely to weigh these parameters with you and design a personalised strategy — never to impose a standard treatment. To learn more about Dr Zeitoun's training and the institutions where he has worked, see the about page.
Frequently asked questions
Comparison: myomectomy vs hysterectomy
| Criteria | Myomectomy | Hysterectomy |
|---|---|---|
| Fertility | ✅ Preserved | ❌ Permanent |
| Recurrence | ~25-30% at 10 years | 0% |
| Hospitalisation | 1-3 days | 2-3 days |
| Recovery | 2-4 weeks | 4-6 weeks |
| Indication | Young woman, desire pregnancy | Disabling symptoms, near menopause |
Source: ACOG 2021, NICE 2024, Donnez J. Hum Reprod Update 2016.

